Whenever analogs come up in pharma strategy, someone usually says: “But that situation wasn’t the same as ours.”
In recent weeks, I’ve spoken with several people wrestling with external dynamics: market access uncertainty and competitor positioning.
In a couple of those discussions, historical examples of other companies’ activities served as a useful starting point, reminding me how often analog case studies come up in strategy work.
That said, experience has also taught me that analogs are only useful when they are handled with a clear sense of their limits.
Analogs should not be used as a lazy copy-and-paste exercise.
Their value is not in saying, “This happened before, so we can copy it,” or “A competitor did X last time, so they will do it again.”
Used well, they give teams a better way to think through uncertainty.
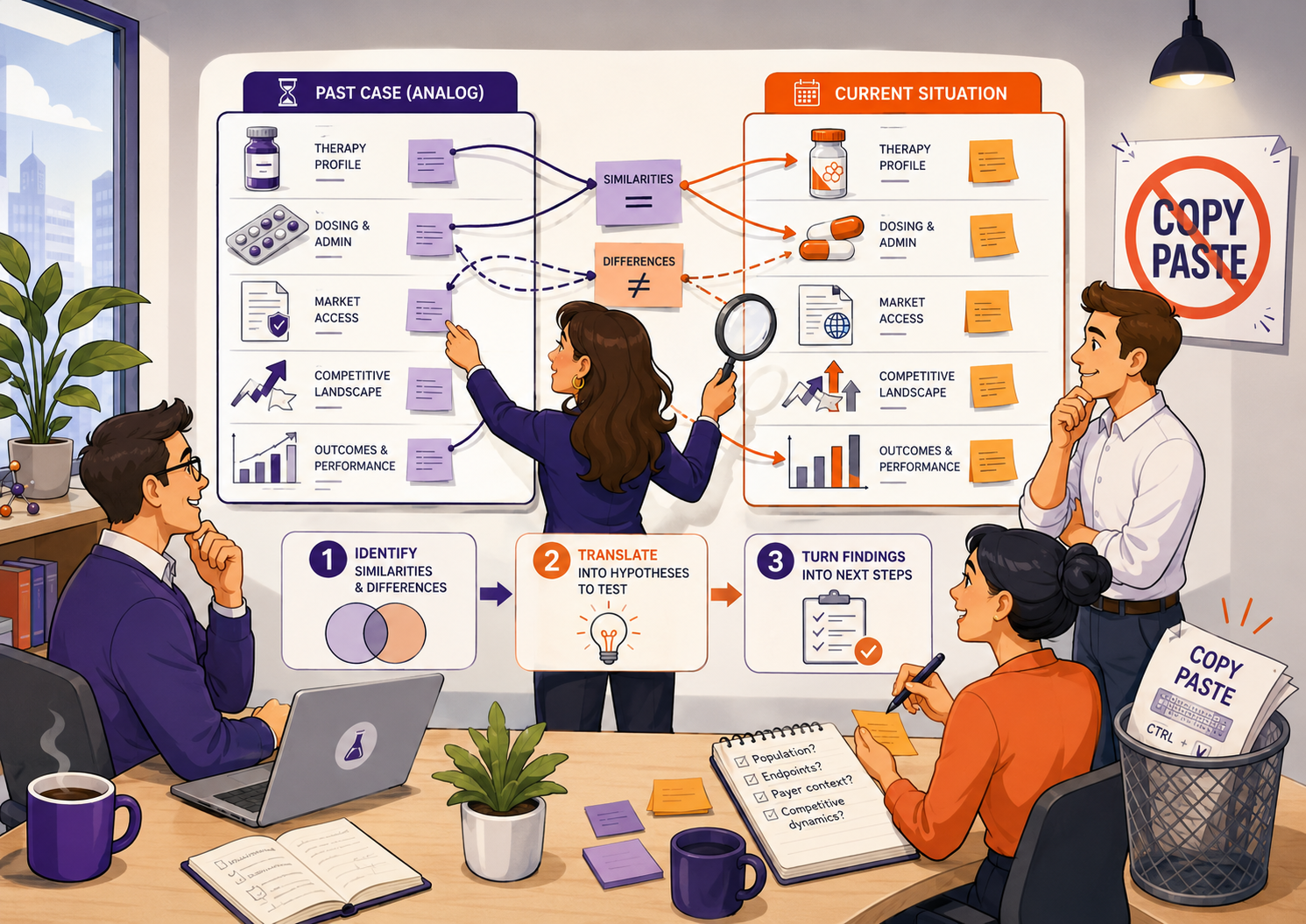
After reviewing an analog case study, I’d suggest using three steps to turn it into something that can support a real decision:
1. Compare the analog properly
What is genuinely similar, and what is meaningfully different? This helps the team work logically through the case, separate what matters from surface-level noise, and avoid getting stuck on the obvious point that “our situation is different.”
2. Translate the analog into testable assumptions
What does the analog suggest we should investigate in our own situation? Create a set of hypotheses or assumptions, prioritise them by importance and uncertainty, then define how to test them: desk research, primary research, expert input, competitor analysis, or internal data.
3. Convert the findings into decisions and actions
What do we do next? Review the findings with the team, decide whether and how to change the strategy, and clarify the activities, resources, owners, and next steps required.
At that point, the analog has done its job: it has moved the team from anecdote to structured decision-making.
The power of an analog is not that it gives you certainty. It is that it gives you a better starting point for uncertainty.