Over recent months, I’ve been exploring novel diagnostic tools, with a particular focus on digital cognitive assessments. Along the way, I came across a Nature Reviews Drug Discovery paper on wearable technologies in clinical trials; a slightly tangential area, but one with plenty of relevance for anyone thinking about evidence generation, patient burden or trial design.
Wearables in clinical trials are often discussed through a technical lens.
What can they measure?
How accurate are they?
Can they support digital endpoints?
Are they validated?
All important questions.
But there is another question that deserves just as much attention:
What is this like for the patient?
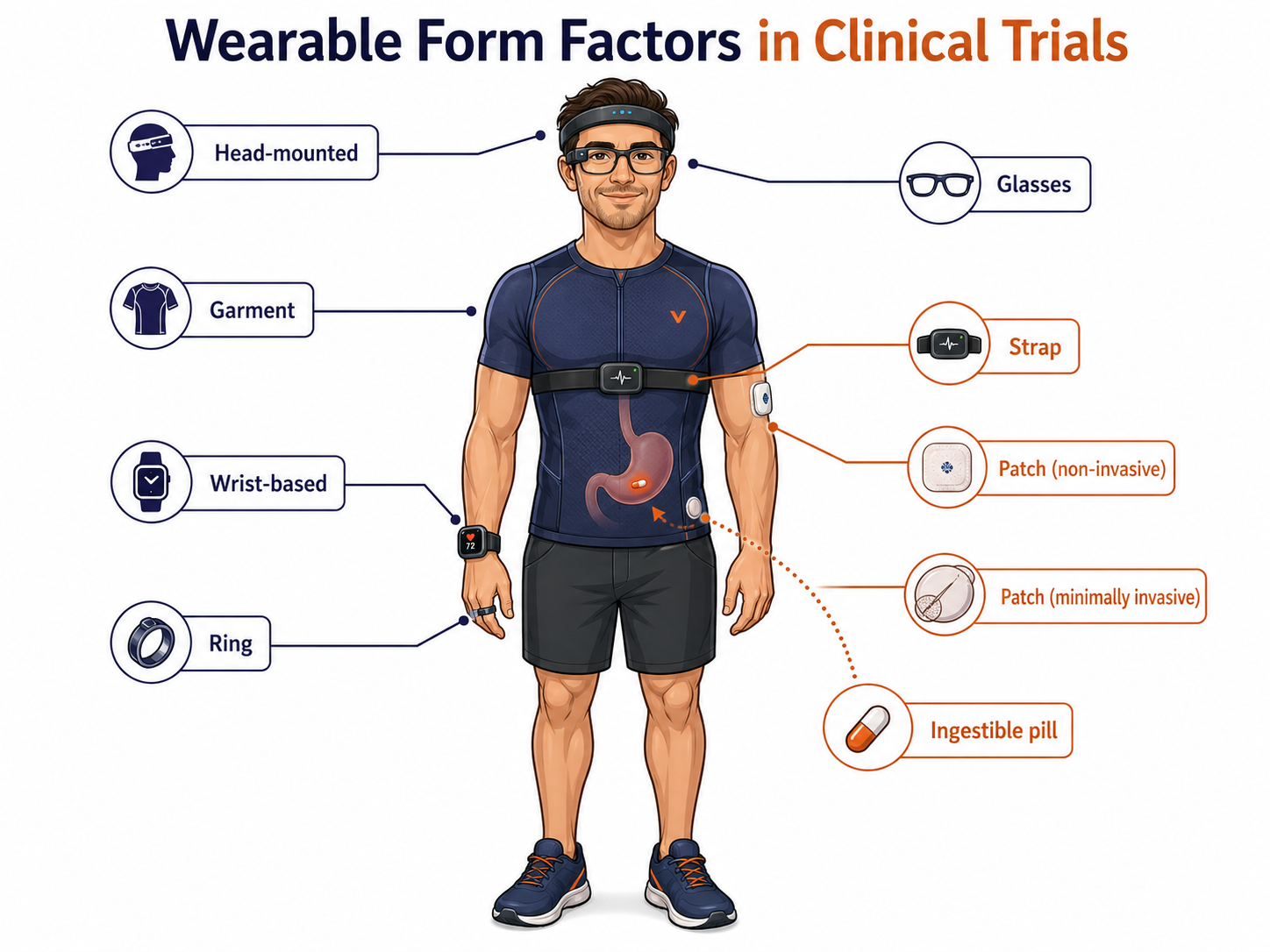
A wrist device may feel familiar.
A patch may feel medical.
A ring may feel discreet.
A head-mounted device may feel like cyborg cosplay.
An ingestible tracking pill may be too invasive.
Each comes with different trade-offs around comfort, discretion, ease of use, anxiety, stigma and day-to-day practicality.
For clinical teams, this affects adherence and data quality.
For medical teams, it affects how evidence is interpreted.
For commercial teams, it affects future differentiation.
For insights teams, it is a reminder that “the patient experience” is rarely a footnote.
I’ve summarised the patient-facing pros and cons across the different wearable form factors the authors identified below, assigning each a Harvey Ball ranking.
Take a read through and let me know your thoughts. If you were taking part in a trial which would you least like to use?
Source: Fayad, Z.A., Hirten, R.P., Nadkarni, G.N. et al. Wearable technologies in clinical trials for drug development: trends and emerging opportunities. Nat Rev Drug Discov (2026). https://doi.org/10.1038/s41573-026-01403-9
Note: Hover over the PDF then use the arrows at the bottom of the viewer to scroll through the pages.
Wearable-Device-Tables_post1